EU Representative Services
The EU Representative acts as a legal interface between the manufacturer and the authorities of the European Union. The European Authorized Representative (E.A.R), located within Europe, must be named by manufacturers who lack a physical European location in Europe. It is required for CE Certification under Medical Device Regulation (MDR) 2017/745 and In Vitro-Diagnostic Medical Device (IVDR) 2017/746. Under the new Medical Device Regulations and Invitro diagnostic device regulations, the EU representative fulfills important duties.
EU Representative Responsibilities
Article 11 of MDR 2017/745 details the important obligations & responsibilities of the European Authorized Representative(EC Rep).
1. There must be a written agreement signed and dated by the manufacturer and the EU Representative.
2. The manufacturer shall enable the representative to perform the below tasks if necessary:
- Review manufactures DOC and CE Certificate and confirms updated Technical File with the manufacturer; verify that an appropriate conformity assessment procedure has been carried out.
- Ensure the Technical File Documentation is available for 10 years (medical devices) and 15 years (implantable devices) after the last device is placed on the market.
- Register on EUDAMED to obtain a single registration number (SRN)
- Pass information to the competent authorities as and when requested or needed.
- Make sure the manufacturer has complied with the Unique Device Identification (UDI) requirements.
- Vigilance monitoring and immediately inform the manufacturer of complaints or reports on devices.
- Terminate the contract if the manufacturer acts contrary to its obligations under the Regulation, and immediately Notified Body
3. The manufacturer cannot delegate third persons to obligations, such as design and manufacture, risk analysis, clinical evaluation, technical documentation, declaration of conformity, or UDI compliance
4. Where the manufacturer is not established in a member state and has not complied with general obligations of the manufacturer (article 10), the A.R. shall be legally liable for defective devices on the same basis as, and jointly and severally with, the manufacturer.
Furthermore, Article 15 of MDR 2017/745 lists obligations of EAR including the necessity of one person responsible for regulatory compliance, who possesses the requisite expertise regarding the regulatory requirements for medical devices in the European Union.
Medical Device Competent Authority registration. (EU Registration)
All medical devices sold in Europe must be registered with the Competent Authority. Registration must be done with the competent authority belonging to your locality. All non-European manufacturers must register with the competent authority where your EC / EU Representative is based.
Class 1 device can be registered after signing the declaration of Conformity (DOC) and CE Mark on the device. In contrast, Class Is, Ir, Im, IIa, IIb, or III devices are placed on their markets after receiving CE Certificate from Notified Body.
European Free Sale Certificate (FSC)
The CE marked product shows conformity to all obligations for medical devices as required by the European community as per Medical Devices Directives. This means you have fulfilled all laws as per EU, EEA, and the few other countries having mutual recognition agreements in place.
However, if you want to sell your items in non-EU countries, you’ll need a Free Sales Certificate. This certificate should be submitted by the manufacturer to the non-European nation that will be marketing your items. The Free Sales Certificate demonstrates from the government that you place products legally with the CE mark on the European market. It’s Just a guarantee!!!
- The Competent Authority of the EU member state where the “manufacturer” in Europe or his authorized agent has his registered place of business can issue the European Free Sales Certificate (FSC) / Certificate of Marketability.
- Only the EU Representative can obtain the Free Sales Certificate on behalf of the manufacturer if the manufacturer is not located in Europe.
Some of the countries that require a Free sale certificate are the following……..
Argentina, Brazil, Chile, China, Colombia, Costa Rica, Ecuador, Egypt, Guatemala, Hong Kong, India, Indonesia, Israel, Mexico, Panama, Peru, Russia, Saudi Arabia, Singapore, Sri Lanka, Syria, Taiwan, Thailand, United Arab Emirates, Venezuela, and Vietnam.
The UDI data will be registered on the EUDAMED database.
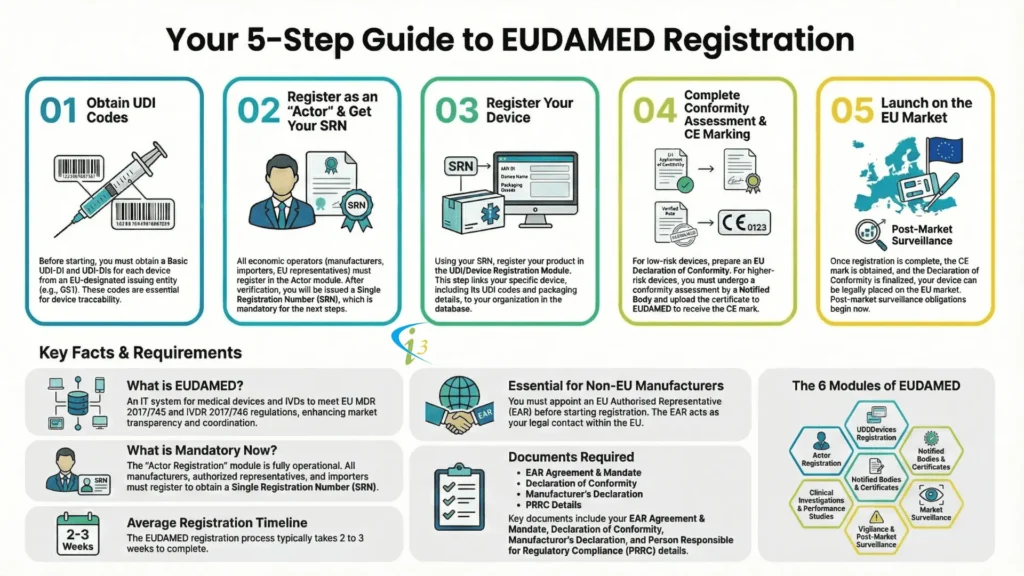
EUDAMED Registration Procedure
Referring to articles 29, 30, and 31, before the devices are subjected to the CA procedure, the manufacturer should assign a basic UDI-DI and provide it to the UDI database. The Notified Body will add a reference of the basic UDI-DI in the certificate issued and will check the EUDAMED whether the information added is correct or not.
The manufacturer should keep the information updated thereafter in the EUDAMED. Now regarding the registration of manufacturers, the competent authority will be providing a Single Registration Number (SRN) to the manufacturer upon the registration at an electronic system managed by the MDCG. The manufacturer should approach the NB with the SRN to access the EUDAMED and for conformity assessment, indicating that both procedures are done before getting the CE certificate.
SRN Number
Every economic operator in and outside of Europe is identified by the Single Registration Number (SRN Number). The SRN is issued once the Actor registration request has been confirmed by the applicable competent body. A single registration number is assigned to all medical device legal manufacturers, EU representatives, system/procedure pack producers, and importers involved in placing medical devices and IVDs on the European market.
Single Registartion Number (SRN) is mandatory for the following type of actors
- Manufacturer (or legal manufacturer)
- EU Authorized Representative
- System and procedure pack producer
- Importer
Above actors must know where is the SRN used. Once a Single Registration Number is issued to a device manufacturer it can be used in various locations such as
- Free Sale Certificate
- Declaration of Conformity (MDR/IVDR)
- Notified Body CE Certificate
- Technical Documentation (MDR/IVDR)

Unique Identification Number (UDI)
The EU MDR mandates the use of a Unique Device Identification (UDI) system, allowing the identification and facilitating the traceability of devices.
It can be implemented by establishing an electronic system for the Unique Device Identification (UDI Database).
- Place the UDI on the label of the device or its packaging
- Make it appear on the EU Declaration of Conformity.
- Should keep up-to-date lists of all the UDIs that have been assigned.
- Register the device – the manufacturer should assign a Basic UDI-DI to the device and provide it to the UDI database and the other core data elements related to that device.
- Register themselves – Before placing a device on the market, manufacturers should register, and submit it to the electronic system if they have not already registered. In cases where the conformity assessment procedure requires the involvement of a notified body, the information shall be provided to that electronic system before applying to the notified body.
Unique Device Identification (UDI) System
Unique Device Identification (UDI) number is a set of alphanumeric or numeric characters created through internationally accepted identification and coding standards that would allow distinct identification and traceability of specific devices in the market. The UDI is comprised of Device Identifier UDI-DI (specific to a manufacturer and a device) and Production Identifier UDI-PI (specific to the unit of device production and the packaged devices). Both Device Identifier (UDI-DI) and a Production Identifier (UDI-PI) elements
UDI-DI
- The primary Identifier of a device model
- provides a link to the UDI database.
- Referenced in relevant certificates and declaration of conformity.
UDI-PI
- Numeric or alphanumeric code that identifies the unit of device production.
- Includes serial & lot number, software ID, and manufacturing & expiry date or both types of data.
- Referenced in relevant certificates and declaration of conformity.
Application of the UDI
- UDI number on the label of the device or its packaging
- Recording of the UDI by economic operators, health institutions, and healthcare professionals.
- Establishment of an electronic system for Unique Device Identification (UDI database).
- The UDI data will be registered on the EUDAMED database.
Who is issuing the UDI – DI?
UDI-DI is issued by the ‘issuing entities’ designated by the EU Commission by means of implementing acts. The manufacturer then assigns the UDI to the device and all higher-level packages (except shipment container) before placing them on the market, complying with the issuing entity’s rules. Some UDI issuing entities are GS1, HIBCC, and ICCBA.
What should manufacturers do for that?
The manufacturer has to apply for the UDI to the issuing entity designated by the EU commission. The UDI has to be submitted to the UDI database with the details of all the packaging levels. The appropriate procedure has to be maintained for the validation of the data. They have to periodically verify the correctness of all the data to the devices placed on the market and available for use.
Any change in the below details of the device requires a new UDI-DI
- name or trade name,
- device version or model
- labeled as single-use
- packaged sterile
- need for sterilization before use
- quantity of devices provided in the package
- critical warnings or contra-indications
Other than the above changes, the device doesn’t require a new UDI-DI, and the manufacturer has to update the UDI database within 30 days of the change being made to the element. The UDI of an implantable device should be at the lowest level of packaging and should be identifiable before implantation. Reusable devices requiring cleaning, disinfection, sterilization, or refurbishing between UDI devices should be placed on the device and readable after each process. Configurable devices should bear configurable device UDI. The systems and procedure pack the UDI carrier be affixed on the outside of the packaging with some exceptions.
How to implement UDI?
The EU commission will designate one or several issuing entities to operate a system for UDI assignment based on the Regulation. The manufacturer has to assign the UDI with the help of the issuing entity.
The assigned UDI has to be submitted to the UDI database by the manufacturer. Any updates in the identifying information and other device data elements have also to be updated in the database. Before placing the devices other than custom-made and investigational devices, the manufacturers have to affix the UDI on the labels or on the device itself and on all higher levels of packaging.
EAR Annual Service Fees (MDR / IVDD)
Timeline for E.A.R Agreement Drafting and Signing: 2 – 3 working days
|
# |
Device Risk Class |
Annual Fees in euros |
Timeline |
| 1 | Class l | 850 | 7-10 days |
| 2 | Class ls, 1m, 1r, IIa | 950 | 7-10 days |
| 3 | Class IIb | 1150 | 7-10 days |
| 4 | Class III | 1350 | 7-10 days |
| 6 | Free Sale Certificate | 3500 | 40-60 days |
| 7 | Competent Authority Registration | 500 | 14-20 days |
| 8 | E.A.R Certificate Attestation from CC | 150 | 7-10 days |
If you have multiple devices and need a customized quote or wish to know more about EU Representative roles under the new MDR Regulation,


Our Social Activities